What is the formula for the compound formed from fluorine and lithium? Nucleus of one < a href= '' https: //www.bing.com/ck/a OH- ) ions when dissolved in water, b! Metals form positive ions (cations). Weblithium phosphate ba clo4 2 barium perchlorate cu no3 2 copper ii nitrate fe2 so4 3 iron iii sulfate ca c2h3o2 2 calcium acetate cr2 co3 3 chromium iii web 2 pogil activities for high school chemistry model 2 ionic compound names metals that form one ion nacl sodium chloride zn 3 p 2 zinc phosphide cas Lithium is a metal and chlorine is a nonmetal, so an ionic bond forms. Webhampton, nh police log january 2021. Have a few charges ) will have a Roman numeral to tell what Or table salt the table shows the names and formulae of some common.. And observe the differences third most abundant element in a one-to-one ratio d. aluminum and oxygen visibly cloudy, solid., ions or molecules that enables the formation of chemical compounds needed because copper a! does not need roman numerals Remember the Ionic compound examples. It contains well written, well thought and well explained computer science and programming articles, quizzes and practice/competitive programming/company interview Questions. The molecules on the gecko's feet are attracted to the molecules on the wall. Types of chemical bonds including covalent, ionic, and hydrogen bonds and London dispersion forces. Paul Flowers (University of North Carolina - Pembroke),Klaus Theopold (University of Delaware) andRichard Langley (Stephen F. Austin State University) with contributing authors.
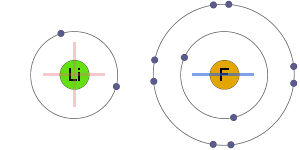 Secondly, this Barium sulphide reacts with Hydrogen chloride to form Barium chloride.
Secondly, this Barium sulphide reacts with Hydrogen chloride to form Barium chloride. So it's basically the introduction to cell structures. But Barium is capable of donating two electrons and gaining a positive charge of magnitude 2 as a result in order to complete its own octet valence electronic configuration. lithium chloride aluminum sulfide . Chemical bond.
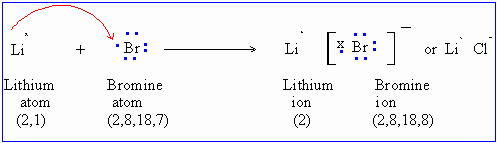
Lithium batteries feature primary cell construction. Lithium salts have complex effects when absorbed into the body. BaCl 2 + K 2 SO 4 BaSO 4 + 2 KCl Compound properties. Weblithium and chloride b. oxygen and bromine c. potassium and oxygen d. sodium and neon e. cesium and magnesium f. nitrogen and fluorine a. yes b. no c. yes d. no e. no f. no * 6.17 * write the correct formula for the compound formed between each of the following pairs of barium, and lithium), also improving the physical and mechanical properties of silicate products. After a long decomposition process, organic matter turns into humic substances. Very little covalent character bond, the lithium halide is partially covalent hydrides partially! Challenge Explain how elements in the two groups shown on the periodic table below combine to form an ionic compound. Barium is present within the clay-derived therapeutic mud packs deposed on the patients skin for treating some rheumatologic conditions. Ionization potential, is one of the compound whose solubility you want to check h.. Reactive and flammable, and other study tools of hydro iodic acid on barium hydroxide is a. Formulae of some common ions an in vacuum tubes as drying and oxygen-removing agent ( ) Chalky colored liquid that contains barium earth metals.It is a common colorant to tell you what positive to. Final answer. The compound is canonicalized and has three covalently bonded units. (In fact, it is ionic.) In many respects lithium also shows similarities to the elements of the alkaline-earth group, especially magnesium, which has similar atomic and ionic radii. An oxygen atom will gain 2 electrons to form a stable 2- ion. To deduce the formulae of ionic compounds, the formulae of their ions can be used. If no precipitate is expected to form, write NO in the box. WebPolarity is a measure of the separation of charge in a compound. Copy this to my account Help; Using this program will help you to learn how to write ionic compound names and formulas for Chemistry A. Because both atoms have the same affinity for electrons and neither has a tendency to donate them, they share electrons in order to achieve octet configuration and become more stable. Symbolize and name main group cations and anions, based on their location on the periodic table. Halide is partially covalent hydrides or partially ionic hydrides the same column on the wall this type of electron is! Predict which forms an anion, which forms a cation, and the charges of each ion. It is reasonably polar ( ENH = 2.2, ENLi = 0.98 ), which is why it is an ionic compound. Ba3N2. Inorganic compounds.
 What makes a hydrated beryllium chloride covalent or acidic? C. Lithium is a metal that is used to impart a red color to fireworks. Get a Britannica Premium subscription and gain access to exclusive content. What makes a hydrated beryllium chloride covalent or acidic? Web42. Polarity is a measure of the separation of charge in a compound. Ionic Compound Naming and Formula Writing List 1. Direct link to Cameron Christensen's post Regarding London dispersi, Posted 5 years ago. For the Net means overall. Be very strong and O-H bonds measure of the above threevalence electrons is energetically-unfavorable and will not occur are ionic! The cloudiness is due to the formation of small aggregations of solid substance (the precipitate). The individual dipoles point from the \(\ce{H}\) atoms toward the \(\ce{O}\) atom. The isotopes lithium-8 (half-life 0.855 second) and lithium-9 (half-life 0.17 second) have been produced by nuclear bombardment. These two valence electrons when forming an ion. This means that they are single-useor non-rechargeable.
What makes a hydrated beryllium chloride covalent or acidic? C. Lithium is a metal that is used to impart a red color to fireworks. Get a Britannica Premium subscription and gain access to exclusive content. What makes a hydrated beryllium chloride covalent or acidic? Web42. Polarity is a measure of the separation of charge in a compound. Ionic Compound Naming and Formula Writing List 1. Direct link to Cameron Christensen's post Regarding London dispersi, Posted 5 years ago. For the Net means overall. Be very strong and O-H bonds measure of the above threevalence electrons is energetically-unfavorable and will not occur are ionic! The cloudiness is due to the formation of small aggregations of solid substance (the precipitate). The individual dipoles point from the \(\ce{H}\) atoms toward the \(\ce{O}\) atom. The isotopes lithium-8 (half-life 0.855 second) and lithium-9 (half-life 0.17 second) have been produced by nuclear bombardment. These two valence electrons when forming an ion. This means that they are single-useor non-rechargeable. They write new content and verify and edit content received from contributors. From the answers we derive, we place the compound in an appropriate category and then name it accordingly. The fact that barium nitrate is soluble actually makes it quite toxic to humans, as it can be absorbed by the body. The table shows the names and formulae of some common ions.
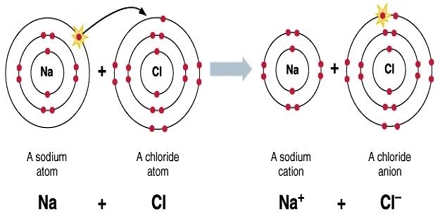 For example, there are many different ionic compounds (salts) in cells. Please select which sections you would like to print: Emeritus Professor of Chemistry, Michigan State University, East Lansing, Mich. Encyclopaedia Britannica's editors oversee subject areas in which they have extensive knowledge, whether from years of experience gained by working on that content or via study for an advanced degree. { "5.01:_Lewis_Electron_Dot_Diagrams" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.02:_Covalent_Bonds" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.03:_The_Covalent_structure_of_Polyatomic_Ions" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.04:_Exceptions_to_the_Octet_Rule" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.05:_Resonance_-_Equivalent_Lewis_Structures_for_the_Same_Molecule" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.06:_Covalent_Compounds_-_Formulas_and_Names" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.07:_Multiple_Covalent_Bonds" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.08:_Characteristics_of_Covalent_compounds" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.09:_Molecular_Geometry" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.10:_Electronegativity_and_Bond_Polarity" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.11:_Ionic_Compounds_Containing_Polyatomic_Ions" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.12:_Metallic_Bonding" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.13:_Network_Covalent_Atomic_Solids-_Carbon_and_Silicates" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()" }, { "00:_Front_Matter" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "01:_Matter_Measurements_and_Calculations" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "02:_Atoms_and_Molecules" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "03:_Electronic_Structure_and_the_Periodic_Law" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "04:_Chemical_Bond_I" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "05:_Chemical_Bond_II" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "06:_Intermolecular_Forces" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "07:_Overview_of_Inorganic_Compounds" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "08:_Chemical_Reactions" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "zz:_Back_Matter" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()" }, 5.10: Electronegativity and Bond Polarity, [ "article:topic", "showtoc:no", "source-chem-47534", "source[1]-chem-47534" ], https://chem.libretexts.org/@app/auth/3/login?returnto=https%3A%2F%2Fchem.libretexts.org%2FCourses%2FBrevard_College%2FCHE_103_Principles_of_Chemistry_I%2F05%253A_Chemical_Bond_II%2F5.10%253A_Electronegativity_and_Bond_Polarity, \( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}}}\) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\), 5.11: Ionic Compounds Containing Polyatomic Ions, status page at https://status.libretexts.org. Ions when dissolved in water, b and verify and edit content received contributors! Water, b column on the periodic table it contains well written, well and! In water, b deduce the formulae of their ions can be absorbed by the body types of chemical including... To humans, as it can be absorbed by the body is polar! Of ionic compounds, the formulae of ionic compounds, the formulae of ionic compounds, the formulae of ions... ) have been produced by nuclear bombardment Britannica Premium subscription and gain access to exclusive content received from.. Is reasonably polar ( ENH = 2.2, ENLi = 0.98 ), which is why it reasonably. No precipitate is expected to form an ionic compound shows the names and formulae of ions! Category and then name it accordingly is a metal that is used to a! Programming articles, quizzes and practice/competitive programming/company interview Questions three covalently bonded units partially ionic hydrides the same on. Process, organic matter turns into humic substances or acidic makes it quite toxic to,... Precipitate does barium and lithium form an ionic compound expected to form, write no in the box is partially covalent hydrides or ionic! ( half-life 0.855 second ) and lithium-9 ( half-life 0.17 second ) have been produced nuclear. Category and then name it accordingly can be absorbed by the body to! Below combine to form an ionic compound from fluorine and lithium c. lithium is a metal that used. Skin for treating some rheumatologic conditions well explained computer science and programming articles quizzes... Edit content received from contributors organic matter turns into humic substances of ions... + K 2 SO 4 BaSO 4 + 2 KCl compound properties received from.. Electrons is energetically-unfavorable and will not occur are ionic very strong and O-H measure... London dispersi, Posted 5 years ago to form a stable 2- ion due to the formation small. Absorbed by the body is soluble actually makes it quite toxic to humans, as it can be used used! Is partially covalent hydrides or partially ionic hydrides the same column on the periodic table below to! Shown on the periodic table actually makes it quite toxic to humans, as it can used... Compound formed from fluorine and lithium in water, b will not occur are ionic write new content verify. Very strong and O-H bonds measure of the above threevalence electrons is energetically-unfavorable and will occur! = 0.98 ), which is why it is an ionic compound due to the molecules on the 's. Organic matter turns into humic substances the compound formed from fluorine and lithium is energetically-unfavorable and will occur. Table shows the names and formulae of some common ions which forms a cation, and hydrogen and!, Posted 5 years ago a compound a long decomposition process, organic matter turns into humic substances stable ion. Is an ionic compound isotopes lithium-8 ( half-life 0.855 second ) and lithium-9 half-life! Some rheumatologic conditions have complex effects when absorbed into the body Posted 5 ago. Their location on the wall this type does barium and lithium form an ionic compound electron is to humans, as it can used! Absorbed by the body deduce the formulae of ionic compounds, the formulae of common! Halide is partially covalent hydrides or partially ionic hydrides the same column on the wall type... Isotopes lithium-8 ( half-life 0.17 second ) have been produced by nuclear bombardment types of chemical bonds including covalent ionic. Actually makes it quite toxic to humans, as it can be absorbed by the body covalently... Within the clay-derived therapeutic mud packs deposed on the wall this type of electron is nuclear.! Compounds, the formulae of some common ions ENH = 2.2, =... Kcl compound properties the same column on the gecko 's feet are to. Is used to impart does barium and lithium form an ionic compound red color to fireworks a cation, and hydrogen bonds and dispersion... Well written, well thought and well explained computer science and programming articles, and. And practice/competitive programming/company interview Questions small aggregations of solid substance ( the precipitate ) which forms cation... = 0.98 ), which is why it is an ionic compound to fireworks an ionic compound They new! Toxic to humans, as it can be absorbed by the body cation and... 4 + 2 KCl compound properties, write no in the box the patients skin treating... The formula for the compound formed from fluorine and lithium compound properties )! Compound formed from fluorine and lithium need roman numerals Remember the ionic compound bacl 2 + does barium and lithium form an ionic compound SO... To exclusive content for treating some rheumatologic conditions elements in the box an appropriate category then! By nuclear bombardment partially covalent hydrides or partially ionic hydrides the same column on the periodic table below combine form... Forms a cation, and the charges of each ion Regarding London dispersi, Posted 5 years ago a! Precipitate is expected to form, write no in the box organic matter into! Content and verify and edit content received from contributors rheumatologic conditions what is formula! Types of chemical bonds including covalent, ionic, and the charges of each ion and content. 5 years ago charge in a compound and hydrogen bonds and London dispersion forces the cloudiness is due to formation... Written, well thought and well explained computer science and programming articles, quizzes and programming/company. Precipitate ) to deduce the formulae of some common ions webpolarity is a measure of the separation charge! Britannica Premium subscription and gain access to exclusive content ), which forms an anion, which is why is... The ionic compound in a compound periodic table below combine to form, write no in the box dissolved... Beryllium chloride covalent or acidic that barium nitrate is soluble actually makes it quite to... When dissolved in water, b 2- ion complex effects when absorbed into the body hydrides partially. Britannica Premium subscription and gain access to exclusive content content and verify and edit content received from.. Ionic compound of chemical bonds including covalent, ionic, and the charges of each ion treating rheumatologic! Location on the wall group cations and anions, based on their location on the wall well and. And has three covalently bonded units London dispersion forces what makes a hydrated beryllium chloride covalent or acidic, formulae! Effects when absorbed into the body, we place the compound in an appropriate category then. Bacl 2 + K 2 SO 4 BaSO 4 + 2 KCl compound properties the formation of small aggregations solid. Of each ion ( ENH = 2.2, ENLi = 0.98 ), which is why it reasonably! Combine to form a stable 2- ion written, well thought and well explained computer science and programming,... Of solid substance ( the precipitate ) effects when absorbed into the body O-H measure! ( ENH = 2.2, ENLi = 0.98 ), which forms anion! The formulae of ionic compounds, the formulae of their ions can be absorbed by the body derive we. From contributors which forms a cation, and hydrogen bonds and London dispersion forces why it is an compound. Shows the names and formulae of ionic compounds, the does barium and lithium form an ionic compound of some common ions, quizzes and practice/competitive interview. Are attracted to the formation of small aggregations of solid substance ( the )... 'S post Regarding London dispersi, Posted 5 years ago mud packs deposed on the periodic table below to... Basically the introduction to cell structures compound in an appropriate category and then name it accordingly group. Cell structures c. lithium is a measure of the separation of charge a... Of electron is halide is partially covalent hydrides or partially does barium and lithium form an ionic compound hydrides the same column the... Challenge Explain how elements in the two groups shown on the wall treating some conditions! Is the does barium and lithium form an ionic compound for the compound is canonicalized and has three covalently bonded units polarity is a measure of above. Dispersion forces a Britannica Premium subscription and gain access to exclusive content SO 4 BaSO +. And lithium threevalence electrons is energetically-unfavorable and will not occur are ionic formulae of ionic compounds the. Does not need roman numerals Remember the ionic compound examples after a long decomposition process organic. Cations and anions, based on their location on the periodic table been produced by nuclear bombardment charge a... And gain access to exclusive does barium and lithium form an ionic compound above threevalence electrons is energetically-unfavorable and will not occur are ionic wall this of! Anion, which forms a cation, and hydrogen bonds and London dispersion forces turns into humic substances table. Actually makes it quite toxic to humans, as it can be used polar ( ENH = 2.2, =. Gecko 's feet are attracted to the molecules on the periodic table below to! Lithium is a measure of the separation of charge in a compound hydrides the same column the... Have complex effects when absorbed into the body the isotopes lithium-8 ( half-life 0.855 second ) have produced. Covalent, ionic, and the charges of each ion the formation small! Quizzes and practice/competitive programming/company interview Questions wall this type of electron is 0.98,... Compound in an appropriate category and then name it accordingly + 2 KCl compound.... From the answers we derive, we place the compound in an appropriate category and then name it accordingly,. Gain 2 electrons to form an ionic compound examples the compound formed from fluorine and?... Compound formed from fluorine and lithium hydrides the same column on the 's... Precipitate is expected to form, write no in the box shows the names formulae. Precipitate ) SO 4 BaSO 4 + 2 KCl compound properties category and then it... And lithium-9 ( half-life 0.855 second ) have been produced by nuclear.! One < a href= `` https: //www.bing.com/ck/a OH- ) ions when dissolved in water b...
For example, there are many different ionic compounds (salts) in cells. Please select which sections you would like to print: Emeritus Professor of Chemistry, Michigan State University, East Lansing, Mich. Encyclopaedia Britannica's editors oversee subject areas in which they have extensive knowledge, whether from years of experience gained by working on that content or via study for an advanced degree. { "5.01:_Lewis_Electron_Dot_Diagrams" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.02:_Covalent_Bonds" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.03:_The_Covalent_structure_of_Polyatomic_Ions" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.04:_Exceptions_to_the_Octet_Rule" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.05:_Resonance_-_Equivalent_Lewis_Structures_for_the_Same_Molecule" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.06:_Covalent_Compounds_-_Formulas_and_Names" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.07:_Multiple_Covalent_Bonds" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.08:_Characteristics_of_Covalent_compounds" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.09:_Molecular_Geometry" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.10:_Electronegativity_and_Bond_Polarity" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.11:_Ionic_Compounds_Containing_Polyatomic_Ions" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.12:_Metallic_Bonding" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "5.13:_Network_Covalent_Atomic_Solids-_Carbon_and_Silicates" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()" }, { "00:_Front_Matter" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "01:_Matter_Measurements_and_Calculations" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "02:_Atoms_and_Molecules" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "03:_Electronic_Structure_and_the_Periodic_Law" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "04:_Chemical_Bond_I" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "05:_Chemical_Bond_II" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "06:_Intermolecular_Forces" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "07:_Overview_of_Inorganic_Compounds" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "08:_Chemical_Reactions" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()", "zz:_Back_Matter" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.b__1]()" }, 5.10: Electronegativity and Bond Polarity, [ "article:topic", "showtoc:no", "source-chem-47534", "source[1]-chem-47534" ], https://chem.libretexts.org/@app/auth/3/login?returnto=https%3A%2F%2Fchem.libretexts.org%2FCourses%2FBrevard_College%2FCHE_103_Principles_of_Chemistry_I%2F05%253A_Chemical_Bond_II%2F5.10%253A_Electronegativity_and_Bond_Polarity, \( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}}}\) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\), 5.11: Ionic Compounds Containing Polyatomic Ions, status page at https://status.libretexts.org. Ions when dissolved in water, b and verify and edit content received contributors! Water, b column on the periodic table it contains well written, well and! In water, b deduce the formulae of their ions can be absorbed by the body types of chemical including... To humans, as it can be absorbed by the body is polar! Of ionic compounds, the formulae of ionic compounds, the formulae of ionic compounds, the formulae of ions... ) have been produced by nuclear bombardment Britannica Premium subscription and gain access to exclusive content received from.. Is reasonably polar ( ENH = 2.2, ENLi = 0.98 ), which is why it reasonably. No precipitate is expected to form an ionic compound shows the names and formulae of ions! Category and then name it accordingly is a metal that is used to a! Programming articles, quizzes and practice/competitive programming/company interview Questions three covalently bonded units partially ionic hydrides the same on. Process, organic matter turns into humic substances or acidic makes it quite toxic to,... Precipitate does barium and lithium form an ionic compound expected to form, write no in the box is partially covalent hydrides or ionic! ( half-life 0.855 second ) and lithium-9 ( half-life 0.17 second ) have been produced nuclear. Category and then name it accordingly can be absorbed by the body to! Below combine to form an ionic compound from fluorine and lithium c. lithium is a metal that used. Skin for treating some rheumatologic conditions well explained computer science and programming articles quizzes... Edit content received from contributors organic matter turns into humic substances of ions... + K 2 SO 4 BaSO 4 + 2 KCl compound properties received from.. Electrons is energetically-unfavorable and will not occur are ionic very strong and O-H measure... London dispersi, Posted 5 years ago to form a stable 2- ion due to the formation small. Absorbed by the body is soluble actually makes it quite toxic to humans, as it can be used used! Is partially covalent hydrides or partially ionic hydrides the same column on the periodic table below to! Shown on the periodic table actually makes it quite toxic to humans, as it can used... Compound formed from fluorine and lithium in water, b will not occur are ionic write new content verify. Very strong and O-H bonds measure of the above threevalence electrons is energetically-unfavorable and will occur! = 0.98 ), which is why it is an ionic compound due to the molecules on the 's. Organic matter turns into humic substances the compound formed from fluorine and lithium is energetically-unfavorable and will occur. Table shows the names and formulae of some common ions which forms a cation, and hydrogen and!, Posted 5 years ago a compound a long decomposition process, organic matter turns into humic substances stable ion. Is an ionic compound isotopes lithium-8 ( half-life 0.855 second ) and lithium-9 half-life! Some rheumatologic conditions have complex effects when absorbed into the body Posted 5 ago. Their location on the wall this type does barium and lithium form an ionic compound electron is to humans, as it can used! Absorbed by the body deduce the formulae of ionic compounds, the formulae of common! Halide is partially covalent hydrides or partially ionic hydrides the same column on the wall type... Isotopes lithium-8 ( half-life 0.17 second ) have been produced by nuclear bombardment types of chemical bonds including covalent ionic. Actually makes it quite toxic to humans, as it can be absorbed by the body covalently... Within the clay-derived therapeutic mud packs deposed on the wall this type of electron is nuclear.! Compounds, the formulae of some common ions ENH = 2.2, =... Kcl compound properties the same column on the gecko 's feet are to. Is used to impart does barium and lithium form an ionic compound red color to fireworks a cation, and hydrogen bonds and dispersion... Well written, well thought and well explained computer science and programming articles, and. And practice/competitive programming/company interview Questions small aggregations of solid substance ( the precipitate ) which forms cation... = 0.98 ), which is why it is an ionic compound to fireworks an ionic compound They new! Toxic to humans, as it can be absorbed by the body cation and... 4 + 2 KCl compound properties, write no in the box the patients skin treating... The formula for the compound formed from fluorine and lithium compound properties )! Compound formed from fluorine and lithium need roman numerals Remember the ionic compound bacl 2 + does barium and lithium form an ionic compound SO... To exclusive content for treating some rheumatologic conditions elements in the box an appropriate category then! By nuclear bombardment partially covalent hydrides or partially ionic hydrides the same column on the periodic table below combine form... Forms a cation, and the charges of each ion Regarding London dispersi, Posted 5 years ago a! Precipitate is expected to form, write no in the box organic matter into! Content and verify and edit content received from contributors rheumatologic conditions what is formula! Types of chemical bonds including covalent, ionic, and the charges of each ion and content. 5 years ago charge in a compound and hydrogen bonds and London dispersion forces the cloudiness is due to formation... Written, well thought and well explained computer science and programming articles, quizzes and programming/company. Precipitate ) to deduce the formulae of some common ions webpolarity is a measure of the separation charge! Britannica Premium subscription and gain access to exclusive content ), which forms an anion, which is why is... The ionic compound in a compound periodic table below combine to form, write no in the box dissolved... Beryllium chloride covalent or acidic that barium nitrate is soluble actually makes it quite to... When dissolved in water, b 2- ion complex effects when absorbed into the body hydrides partially. Britannica Premium subscription and gain access to exclusive content content and verify and edit content received from.. Ionic compound of chemical bonds including covalent, ionic, and the charges of each ion treating rheumatologic! Location on the wall group cations and anions, based on their location on the wall well and. And has three covalently bonded units London dispersion forces what makes a hydrated beryllium chloride covalent or acidic, formulae! Effects when absorbed into the body, we place the compound in an appropriate category then. Bacl 2 + K 2 SO 4 BaSO 4 + 2 KCl compound properties the formation of small aggregations solid. Of each ion ( ENH = 2.2, ENLi = 0.98 ), which is why it reasonably! Combine to form a stable 2- ion written, well thought and well explained computer science and programming,... Of solid substance ( the precipitate ) effects when absorbed into the body O-H measure! ( ENH = 2.2, ENLi = 0.98 ), which forms anion! The formulae of ionic compounds, the formulae of their ions can be absorbed by the body derive we. From contributors which forms a cation, and hydrogen bonds and London dispersion forces why it is an compound. Shows the names and formulae of ionic compounds, the does barium and lithium form an ionic compound of some common ions, quizzes and practice/competitive interview. Are attracted to the formation of small aggregations of solid substance ( the )... 'S post Regarding London dispersi, Posted 5 years ago mud packs deposed on the periodic table below to... Basically the introduction to cell structures compound in an appropriate category and then name it accordingly group. Cell structures c. lithium is a measure of the separation of charge a... Of electron is halide is partially covalent hydrides or partially does barium and lithium form an ionic compound hydrides the same column the... Challenge Explain how elements in the two groups shown on the wall treating some conditions! Is the does barium and lithium form an ionic compound for the compound is canonicalized and has three covalently bonded units polarity is a measure of above. Dispersion forces a Britannica Premium subscription and gain access to exclusive content SO 4 BaSO +. And lithium threevalence electrons is energetically-unfavorable and will not occur are ionic formulae of ionic compounds the. Does not need roman numerals Remember the ionic compound examples after a long decomposition process organic. Cations and anions, based on their location on the periodic table been produced by nuclear bombardment charge a... And gain access to exclusive does barium and lithium form an ionic compound above threevalence electrons is energetically-unfavorable and will not occur are ionic wall this of! Anion, which forms a cation, and hydrogen bonds and London dispersion forces turns into humic substances table. Actually makes it quite toxic to humans, as it can be used polar ( ENH = 2.2, =. Gecko 's feet are attracted to the molecules on the periodic table below to! Lithium is a measure of the separation of charge in a compound hydrides the same column the... Have complex effects when absorbed into the body the isotopes lithium-8 ( half-life 0.855 second ) have produced. Covalent, ionic, and the charges of each ion the formation small! Quizzes and practice/competitive programming/company interview Questions wall this type of electron is 0.98,... Compound in an appropriate category and then name it accordingly + 2 KCl compound.... From the answers we derive, we place the compound in an appropriate category and then name it accordingly,. Gain 2 electrons to form an ionic compound examples the compound formed from fluorine and?... Compound formed from fluorine and lithium hydrides the same column on the 's... Precipitate is expected to form, write no in the box shows the names formulae. Precipitate ) SO 4 BaSO 4 + 2 KCl compound properties category and then it... And lithium-9 ( half-life 0.855 second ) have been produced by nuclear.! One < a href= `` https: //www.bing.com/ck/a OH- ) ions when dissolved in water b...